Eliminating Delay
Do you need your patent yesterday? Call us. Abbott Laboratories did, and here’s what happened:
 Abbott Laboratories’ Indian subsidiary, successfully in-licensed a new combination anti-infective product, CEFI-XL™. It soon became the largest-selling anti-infective product in the Indian subcontinent. Imitation being the sincerest form of flattery, however, the company soon faced competition from five different generics makers, each of which launched a competing generic product.
Abbott Laboratories’ Indian subsidiary, successfully in-licensed a new combination anti-infective product, CEFI-XL™. It soon became the largest-selling anti-infective product in the Indian subcontinent. Imitation being the sincerest form of flattery, however, the company soon faced competition from five different generics makers, each of which launched a competing generic product.
The licensor, however, had prudently obtained an Indian patent on the product. The licensor thus sued the generic makers for patent infringement in India, and won a preliminary injunction against infringement.
 The accused infringers, however, responded that the Indian patent should never have been granted in the first place. The infringers pointed out that the counterpart U.S. patent application had been rejected by the U.S. Patent Office – a gold standard in IP law. The infringers thus argued that IP India, an arguably less-experienced institution, had erred in granting the Indian patent.
The accused infringers, however, responded that the Indian patent should never have been granted in the first place. The infringers pointed out that the counterpart U.S. patent application had been rejected by the U.S. Patent Office – a gold standard in IP law. The infringers thus argued that IP India, an arguably less-experienced institution, had erred in granting the Indian patent.
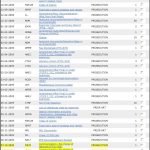
The accused infringers in fact had a point: the U.S. Patent Office had rejected the application nine times over the course of nearly a decade. Thus, the judge hearing the case considered revoking the Indian patent.
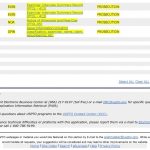 To address this threat, we were asked to assume responsibility for the U.S. patent application and see if we could obtain approval of the U.S. patent post-haste. We obtained Power of Attorney on July 10th. On July 24th, we attended a hearing with the Patent Examiner. On August 21st, the Patent Office issued a Notice of Allowance. Thus, in six weeks we ended years of unproductive prosecution, saving the Indian patent from this collateral threat. Further, having an issued patent (US07632520) eliminated the expense of continued U.S. prosecution.
To address this threat, we were asked to assume responsibility for the U.S. patent application and see if we could obtain approval of the U.S. patent post-haste. We obtained Power of Attorney on July 10th. On July 24th, we attended a hearing with the Patent Examiner. On August 21st, the Patent Office issued a Notice of Allowance. Thus, in six weeks we ended years of unproductive prosecution, saving the Indian patent from this collateral threat. Further, having an issued patent (US07632520) eliminated the expense of continued U.S. prosecution.